A University of Oxford-led study has found that diverse communities of resident commensal gut bacteria collectively protect the human gut from disease-causing microorganisms by consuming the nutrients that the pathogen would need to be able to gain a foothold in the host.
The team used an ecological approach to investigate how colonization by two major bacterial pathogens—Klebsiella pneumoniae and Salmonella enterica serovar Typhimurium (S. Typhimurium)—is influenced by a range of human gut bacteria both in vitro and in gnotobiotic mice. They found that the ecological diversity of the microbiome was important for colonization resistance. While single species of gut microbiota alone had negligible effects on providing effective resistance to pathogens, certain combinations of essential key species within diverse communities exhibited much greater colonization resistance when together.
Having found that these communities block pathogen growth by consuming nutrients that would be required by the pathogen, the team also showed that the concept of nutrient blocking can be used to predict specific sets of commensal microbiota that will resist a novel bacterial pathogen, using genome sequence data alone.
Their combined findings highlight why microbiome diversity is important to human health, and could point to the development of new strategies for optimizing gut health, and for the rational design of pathogen-resistant microbiome communities. Research lead Kevin Foster, PhD, at the departments of biology and biochemistry, University of Oxford, and colleagues reported on their findings in Science, in a paper titled “Microbiome diversity protects against pathogens by nutrient blocking.”
The human gut is home to a diverse community of commensal bacterial species collectively known as gut microbiota. A major health benefit these communities provide is to protect the gut against invading pathogens (disease-causing microorganisms) that could cause harmful infections. “A major health benefit provided by the gut microbiota is protection against pathogen colonization and subsequent infection; a phenomenon known as colonization resistance,” the team noted.
However, while the importance of microbiota for colonization resistance is well known, the microbiome is an immensely diverse and complex ecological system, and this complexity makes understanding which bacterial species and interactions drive colonization resistance against a given pathogen a real challenge. To date, it’s been unclear how the protective effect comes about, and whether certain bacterial species have a more important role than others. “… although the importance of the microbiota for colonization resistance is clear, we currently lack the principles needed to predict, a priori, which microbiota species will be effective against a given pathogen,” the investigators continued. “The constituent species can also affect each other and interact ecologically in ways that are critical for colonization resistance. This combination of species diversity and the potential for ecological interactions makes colonization resistance a challenging phenotype to understand.”
To further investigate how, and which gut microbiota protect against microbial pathogens, the University of Oxford researchers tested 100 different gut bacteria strains individually and in combination for their ability to limit the growth of two harmful bacterial pathogens, Klebsiella pneumoniae and Salmonella enterica. The results showed that while individual gut bacteria showed a very poor ability to restrict the spread of either pathogen, when communities of up to 50 species were cultured together, the pathogens grew up to 1000 times less effectively than when cultured with any individual species.
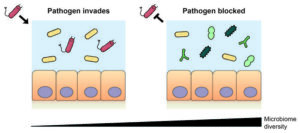
This “community protection effect” was seen regardless of whether the bacteria were cultured together in vials, or in “germ-free” gnotobiotic mice that had no resident gut bacteria at the start of the experiments. “These results clearly demonstrate that colonization resistance is a collective property of microbiome communities,” Foster said. “In other words, a single strain is protective only when in combination with others.”
However, the researchers found that specific members of the bacterial communities—and not just the overall diversity—had a critical effect on the level of protection. Certain microbiome species were found to be essential for community-based protection, even though these species offered little protection on their own.
The researchers demonstrated that protective bacterial communities block pathogen growth by consuming the nutrients that the pathogen needs. As they wrote in their report, “Despite this complexity, we find that these ecological patterns are explained by a simple underlying principle: the collective ability of certain communities to consume nutrients and block pathogen growth … The nutrient-blocking effect is a property of the entire community rather than of any one species alone. That colonization resistance is a community-level trait explains the importance of the ecological diversity and complexity that we observed in our experiments.”
By assessing the genomes of the different bacterial species, the scientists in addition found that the most protective communities were composed of species with highly similar protein compositions to the pathogenic species. “Our genomic analyses suggest that communities that overlap highly with the pathogens in encoded functions provide the best colonization resistance,” they commented.
Metabolic profiling demonstrated that the protective species also had similar demands for carbon sources as the pathogens. “Colonization resistance was only observed once communities shared sufficiently high overlap in their carbon source utilization profile with a pathogen,” the team continued. “Moreover, communities with the greatest metabolic overlap with a pathogen provided the greatest colonization resistance.”
First author Frances Spragge, at the departments of biology and biochemistry, University of Oxford, commented, “Crucially, although increased microbiome diversity increases the probability of protection against these pathogens, the overlap in nutrient utilization profiles between the community and the pathogen is key. Certain species that have a crucial role in community protection show a high degree of metabolic overlap with the pathogen, and therefore similar nutrient demands.”
The researchers used this nutrient blocking principle to predict communities of bacteria that would offer weak and strong protection against an antimicrobial resistant (AMR) E. coli strain. When tested experimentally, the communities that had the highest nutrient overlap with the E. coli strain were up to 100-fold more effective at reducing the pathogen’s abundance than the communities predicted to give weak protection. “We applied our findings to successfully predict communities that resist a novel target strain,” they wrote.
According to the researchers, these new insights could be developed into novel strategies to combat harmful gut pathogens through optimizing gut microbiome communities. The findings may also explain why individuals can become more susceptible to species such as K. pneumoniae after taking antibiotic treatments that can lower the diversity of gut microbiome species.
The post Diverse Gut Microbiomes Block Pathogen Colonization by Consuming Required Nutrients appeared first on GEN – Genetic Engineering and Biotechnology News.