Researchers from Brigham and Women’s Hospital have designed microdevices that can be loaded with drugs and inserted into the brain tumors of patients undergoing standard-of-care surgery, offering a real-time method for measuring the efficacy of drugs against a patient’s specific cancer. The drug-releasing intratumoral microdevices (IMDs)—each the size and shape of a grain of rice—were tested in a small Phase I trial in six patients with high-grade gliomas, demonstrating that they were safe and could be easily integrated into surgical routines.
With further work, the microdevices could feasibly be adopted into clinical and surgical workflows for brain tumors and other cancers to guide treatment decisions and facilitate patient-tailored drug regimens.
“In order to make the greatest impact on how we treat these tumors, we need to be able to understand, early on, which drug works best for any given patient,” said co-principal investigator Pier Paolo Peruzzi, MD, PhD, an assistant professor in the department of neurosurgery at Brigham and Women’s Hospital. “The problem is that the tools that are currently available to answer this question are just not good enough. So we came up with the idea of making each patient their own lab, by using a device which can directly interrogate the living tumor and give us the information that we need.”
Peruzzi is co-corresponding author of the team’s published report in Science Translational Medicine, which is titled “Intratumoral drug-releasing microdevices allow in situ high-throughput pharmaco phenotyping in patients with gliomas.” In their paper, the investigators concluded, “With this first-in-human pilot trial, we provide evidence of safety and feasibility for the use of intratumoral, drug-releasing microdevices as an approach to characterize and compare the efficacy of different pharmacologic therapies in patients with gliomas, in a personalized manner.”
About 20,000 people in the U.S. each year are diagnosed with gliomas, a type of tumor that affects the brain and spinal cord. Gliomas are also among the deadliest brain cancers and are notoriously difficult to treat. One challenge in developing targeted therapies for glioma is that it can be difficult to test many different combinations of drugs in tumor cells, because it’s only possible to treat patients with one approach at a time. This has been a significant barrier for hard-to-treat cancers like gliomas, for which combination therapies are a promising avenue. And, as the authors pointed out, there is a lack reliable methods to predict how well particular cancer drugs will do against tumors, especially brain cancers like glioblastoma. “The lack of reliable predictive biomarkers to guide effective therapy is a major obstacle to the advancement of therapy for high-grade gliomas, particularly glioblastoma (GBM), one of the few cancers whose prognosis has not improved over the past several decades.”
Peruzzi worked closely with co-principal investigator Oliver Jonas, PhD, an associate professor in the department of Radiology at the Brigham, to develop a device that can work around some of the barriers to precision medicine in gliomas. These microdevices are implanted in a patient’s tumor during surgery and removed before the surgery is complete.
“It’s important that we are able to do this in a way that best captures the features of each patient’s tumor and, at the same time, is the least disruptive of the standard of care,” said Peruzzi. “This makes our approach easy to integrate into patients’ treatment and allows its use in real life.”
Over the period of time that the device is implanted—about two to three hours—it administers tiny doses—“nanodoses” of up to 20 drugs into extremely small areas of the patient’s brain tumor. The device is removed at the end of surgery, and the surrounding tissue is returned to the lab for analysis.”
Because the device works while the tumor is still in the body, conducting experiments this way gives unparalleled ability to assess the effects of drugs on the tumor microenvironment, the cells immediately surrounding cancer cells that can make up almost half the mass of a tumor. “This is not in the lab, and not in a petri dish,” said Peruzzi. “It’s actually in real patients in real time, which gives us a whole new perspective on how these tumors respond to treatment.”
For their reported Phase I study the researchers tested their device on six patients undergoing brain surgery to remove a glioma tumor. During their surgeries, each patient received an IMD that released several cancer drugs over the course of the three-hour duration of surgery, at the end of which the IMD, and the remaining piece of tumor in which it was placed, were removed, and the tumor tissue sent to the lab for analysis. “Our approach is based on tiny (6 mm by 0.7 mm) biocompatible intratumoral microdevices (IMDs) that are inserted into the tumor at the beginning of surgery and remain in place while the bulk of the tumor is resected to finally be removed together with the last piece of tumor at the end of surgery,” the team explained. “Each of the pharmaceutical agents loaded into the IMD reservoirs was released upon implantation into a confined region of the tumor directly adjacent to its reservoir.”
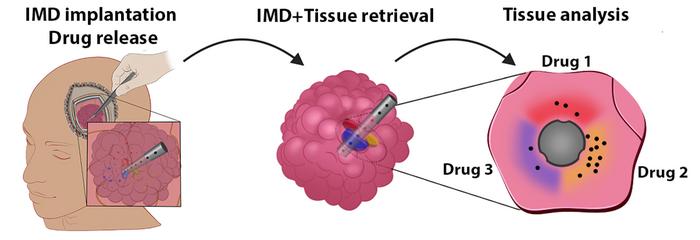
The microdevices were found to be safe and had a very low footprint on the surgery’s performance, adding only 32 minutes to the three-hour procedure. None of the patients experienced any adverse effects from the device, and the researchers were able to collect valuable biological data from the devices, such as how the response changes based on drug concentrations, or molecular changes each drug produces in the cells, and drug-related changes to gene expression in the treated tumor tissue.
In particular, the scientists were able to build detailed profiles about how each patient’s tumor responded to the approved drug temozolomide (TMZ) on the molecular level. A number of patients in the trial received temozolomide as standard anticancer therapy either before, or after the IMD study, so although the trial wasn’t designed to select anticancer agents based on the IMD data, the investigators were able to compare the clinical response to systemic TMZ given as chemotherapy, with the patient-specific response to the drug in the IMD-exposed tissue.
“The intrasurgical use of IMDs in patients with gliomas represents a feasible and promising approach that addresses the need to maximize the efficacy of pharmacotherapy and to understand drug mechanisms of action in the most representative and predictive model,” they concluded. Based on their promising results, the noted that they are currently evaluating the safety of a similar procedure done in two stages over 72 hours, which may maximize data about a drug’s efficacy over longer exposures.
While the study demonstrated that the device was safe and could be easily incorporated into surgical practice, the researchers are still working on determining the exact ways the data it gathers should be used to optimize glioma therapy. The researchers are currently conducting a two-stage version of their procedure in which patients receive the device through minimally invasive surgery 72 hours before their main surgery. And as they noted in their published paper, “Larger clinical studies will be needed to confirm the predictive capability of the IMD to identify systemic responders and to quantitatively define exact thresholds of IMD response correlating with favorable clinical outcomes.”
Nevertheless, they noted, “These results support the potential usefulness of integrating the data obtained from IMDs in the decisional algorithm of an effective, fully personalized adjuvant pharmacologic therapy for these patients.” Peruzzi added, “We’re optimistic that this is a new generation approach for personalized medicine. The ability to bring the lab right to the patient unlocks so much potential in terms of the type of information we can gather, which is new and exciting territory for a disease that has very few options at present.”
The authors further commented, “In addition to providing the ability to directly test a range of drugs in a patient, the use of IMDs in gliomas offers opportunities to answer questions that so far have been elusive.” For example, they said, “… by mixing different drugs into each reservoir, this technology will allow in the future to safely and rapidly test the efficacy of drug combinations, as we have already demonstrated in a preclinical setting.”
Analyzing microdevice-exposed specimens will also give researchers “a realistic vantage point” into the tumor microenvironment, particularly how drugs also affect the non-cancerous cells, such as immune cells, astrocytes, and neurons. And by measuring the drug gradients in a specimen after release from the IMD, scientists may better be able to quantify the tissue concentration at which each drug is biologically effective against the tumor.
The post Human Brain Tumor Implant Could Guide Personalized Therapies appeared first on GEN – Genetic Engineering and Biotechnology News.