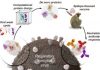
In a recent preprint posted to the SSRN* preprint server, researchers performed a network-based analysis of micro-ribonucleic acids (miRNAs) linked to cognition and Alzheimer’s disease (AD).
 Study: miR-129-5p as a Biomarker for Pathology and Cognitive Decline in Alzheimer’s Disease. Image Credit: ART-ur/Shutterstock.com
Study: miR-129-5p as a Biomarker for Pathology and Cognitive Decline in Alzheimer’s Disease. Image Credit: ART-ur/Shutterstock.com

 *Important notice: SSRN publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: SSRN publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Background
AD pathogenesis remains unclear due to the complicated genetic and neurochemical factors. Dysregulated miRNA expression could contribute to changes in the expression of AD-related genes.
MiRNAs have emerged as promising therapeutic targets due to their crucial role in regulating target gene expression levels. Integrating network analyses and artificial intelligence could provide valuable insights into Alzheimer’s disease pathogenesis and prognostic biomarkers.
About the study
In the present study, researchers explored miRNAs as biomarkers of AD pathogenesis.
The researchers analyzed micro RNAs in the brain tissues of the Religious Orders Study/Rush Memory and Aging Project participants (ROS/MAP, 702 individuals) as the discovery dataset, followed by co-expression network-type analyses to identify network modules of AD dementia and associated miRNA, as well as investigate their relationships with cognition and neuropathological markers.
Brain tissue RNA sequencing (RNA-Seq) data was used to perform association analyses of AD-related micro RNAs and their genetic targets, followed by differential expression analyses of the target genes. In addition to ROS/MAP data, the Gene Expression Omnibus (GEO) data was used to perform consensus co-expression-type network analyses of replicative miRNAs.
Furthermore, the scientists assessed the effectiveness of hub miRNAs for AD categorization using an artificial intelligence-based technique. Gene-set enrichment analysis was performed to identify the biological pathways of target genes strongly related to the expression levels of AD-associated hub miRNAs.
Pathways from the Kyoto Encyclopedia of Genes and Genomes (KEGG) and the Gene Ontology (GO)-biological processes (BP) were utilized. Logistic regression modeling was performed for the analysis.
ROS/MAP participants were classified into those with no cognitive impairments (NCI) and AD. To better differentiate between NCI and AD using neuropathology and clinical criteria, Alzheimer’s diagnosis was based on Braak NFT scores of 4.0 or more, Consortium to Establish a Registry for AD (CERAD) scores, and cognitive probable AD diagnosis without any other cause.
Braak scores of 3.0 or below, CERAD scores denoting potential or no AD, and a clinical NCI diagnosis were used to define NCI.
Alzheimer’s disease was described in Gene Expression Omnibus data obtained from the Brazilian Aging Brain Study Group’s Human Brain Bank, as Braak NFT scores of ≥3.0, and no cognitive impairment as participants without neurological symptoms and neuropathological lesions.
The Accelerating Medicines Partnership for AD (AMP-AD) Portal was used to obtain MiRNA profiles of ROS/MAP participants. Micro RNAs were isolated from the prefrontal cortex on the dorsolateral side of the brain for analysis.
Reverse transcription polymerase chain reaction (RT-PCR) was performed to verify the microarray-identified miRNAs. MiRNA profiles for the GEO dataset were obtained from the National Center for Biotechnology Information (NCBI).
Results
The ROS/MAP cohorts with miRNA included 702 individuals, including 102 NCI and 177 AD patients, with a median age at death of 89 years and 64% females.
The GEO dataset included 16 individuals with miRNA data, including eight NCI individuals and eight AD patients, with a median age of 82 years at death and 69% females.
The network analysis significantly related the glucose metabolism-related module to cognition and AD. In particular, miR-129-5p, miR-1260, miR-433, miR-221, and miR-200a demonstrated robust relationships with the clinicopathological features of Alzheimer’s disease, of which miR129-5p showed the strongest association for all phenotypes.
These five AD-associated hub miRNAs’ target genes were found to be enriched in the erythroblastic leukemia viral oncogene homolog (ErbB), mitogen-activated protein kinase (MAPK), autophagy, 5′ adenosine monophosphate-activated protein kinase (AMPK), and mammalian target of rapamycin (mTOR) signaling pathways.
Notably, miR-129-5p and miR-221 were discovered to be hub miRNAs in the replication cohort after being identified as AD-associated hub miRNAs in the discovery cohort.
Consensus network analysis identified two AD-related consensus network-type modules and two hub micro RNAs (miR-221 and miR-129-5p).
The machine learning investigation revealed that five AD-associated miRNAs significantly enhanced Alzheimer’s disease classification [area under the curve (AUC), 0.8], using covariates such as apolipoprotein E4 (APOE 4) carrier status, sex, and age.
A higher level of miR-129-5p was linked to a decreased risk of Alzheimer’s disease, better cognition, slower cognitive decline, and lower CERAD and Braak pathologic features of Alzheimer’s disease.
Studies have reported that miR-129-5p is associated with AD-related autophagy, neuroinflammation, and neuronal cell death by targeting genes such as the amyloid precursor protein (APP), yes-associated protein 1 (YAP1), and high-mobility group box 1 (HMGB1).
Conclusion
Overall, the study findings highlighted miRNA signatures are associated with AD-related neuropathology and cognition, particularly miR-129-5p.
Integrating network analyses and artificial intelligence could improve Alzheimer’s disease classification and aid in identifying potential biomarkers to develop tailored strategies and enhance the standard of care for AD patients.

 *Important notice: SSRN publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: SSRN publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.