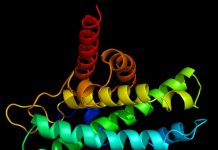
Sub-cellular organelles continue to live and maintain a physical presence, after the cells within which they exist die, a team of scientists from the University of Bristol report. This finding overturns earlier assumptions that organelles decay too quickly to form fossils and compels paleobiologists to take fossilized organelles seriously.
In an article titled “Experimental taphonomy of organelles and the fossil record of early eukaryote evolution” published in Science Advances on January 27, the authors document the decay process of eukaryotic red and green algal cells, showing that nuclei, chloroplasts, and pyrenoids (organelles found within chloroplasts) can persist for weeks and months after the eukaryotic cell dies.
How and when nucleated (eukaryotic) life evolved is contentious because fossil records are difficult to interpret. One way to ascertain the evolutionary timing and assembly of eukaryotes would be to identify eukaryote-grade fossils through fossilized organelles. But claims of fossilized organelles have been discounted in the past, on the grounds that they decay too quickly to undergo fossilization.
The new study on the process of fossilization (taphonomy) characterizes the pattern and time scale of the decay of sub-cellular organelles—nuclei, chloroplasts, pyrenoids—in red and green algae. The evidence shows that these organelles live on for weeks after the death of the organism, a time scale that would allow their preservation through known mechanisms of fossil formation.
Of course, all organelles are not born equal. Or rather, all organelles do not die and decay at the same time as others. Nuclei decay quicker than chloroplasts. Pyrenoids decay even quicker but leave behind spaces within fossilized chloroplasts from which their presence could be inferred.
This taphonomic study demonstrates that claims of fossil organelles can no longer be dismissed. The study is designed not to simulate fossil formation but to estimate whether eukaryotic organelles persist after death as physical entities on a timescale that is compatible with the chemical processes of mineral deposition that leads to the fossilization of organic remains. The study shows that fossilization mechanisms such as phosphatization and silicification can happen within weeks of death.

[Source: Emily Carlisle, Shuhai Xiao]
This prompts the reevaluation and reinterpretation of early eukaryotic fossil records that may make it possible to understand the evolutionary assembly of eukaryotic life.
Emily Carlisle, a PhD student from Bristol’s School of Earth Sciences and co-author, was able to characterizes the decay process of the organelles. “I spent several weeks photographing algal cells as they decayed, checking the condition of the nuclei, chloroplasts and pyrenoids. From this, we could tell that these organelles don’t decay immediately after cell death, but actually take many weeks to dissolve,” she says.
Two billion years after life emerged on earth in the form of simple bacteria, complex life emerged in the form of large, nucleated cells (eukaryotic cells) with membrane-bound organelles, such as chloroplasts and mitochondria. The evolution of fungi, plants and animals followed.
Previous genomic studies suggest eukaryotes may have evolved anywhere from 800 million to 1,800 million years ago. This timeline can be narrowed down considerably using fossil records.
“The evolution of eukaryotes was a hugely important event in the history of life on Earth, but fossils of these cells are difficult to interpret,” said Phil Donoghue, PhD, professor of molecular palaeobiology and one of the co-authors of the study. “Some of them have structures that could be organelles, but there’s long been this assumption that organelles cannot be preserved because they would decay too quickly.”
Early eukaryotes were predominantly single cells, and are difficult to distinguish from bacterial cells in fossils, unlike living eukaryotes that are easily spotted. Conventionally, large size and intricate cell walls are used to identify early eukaryotes. But this can lead to errors as some bacteria can attain large sizes, and cell wall intricacies may be lost to the ravages of time and erosion.
Organelles such as nuclei and chloroplasts are not found in bacteria and would therefore be a better and more conclusive indicator of eukaryotic life forms in fossil records. But organelles have been assumed to decay too quickly to form fossils, without strong evidence to support this assumption.
Data from these experiments shed light on the controversial fossils of early eukaryotic life that include structures within the cells. John Cunningham, PhD and a co-author on the study says, “The structures in Shuiyousphaeridium, a fossil from 1,700 million years ago, closely resemble nuclei. This interpretation has previously been dismissed because of the assumed rapid decay of nuclei. Our decay experiments have shown that nuclei can persist for several weeks, meaning the structures in Shuiyousphaeridium are likely to be nuclei.”
By revealing the decay patterns of organelles, the authors say they can demonstrate the presence of eukaryotic life to 1,700 million years ago, narrowing down their timeline of origin.
The post New Clue on How and When Cells with Nuclei First Evolved appeared first on GEN – Genetic Engineering and Biotechnology News.